
Evidence suggests that the use of a central IRB that oversees multiple sites at the same time significantly expedites the IRB approval process. IRB SubmissionsĪt the site level, IRB submission delays are related to the use of a local IRB. This is especially true if the CRO has utilized a site in the recent past, as they can refer to the previously negotiated contract and budget terms to significantly reduce the timeline for contract execution. 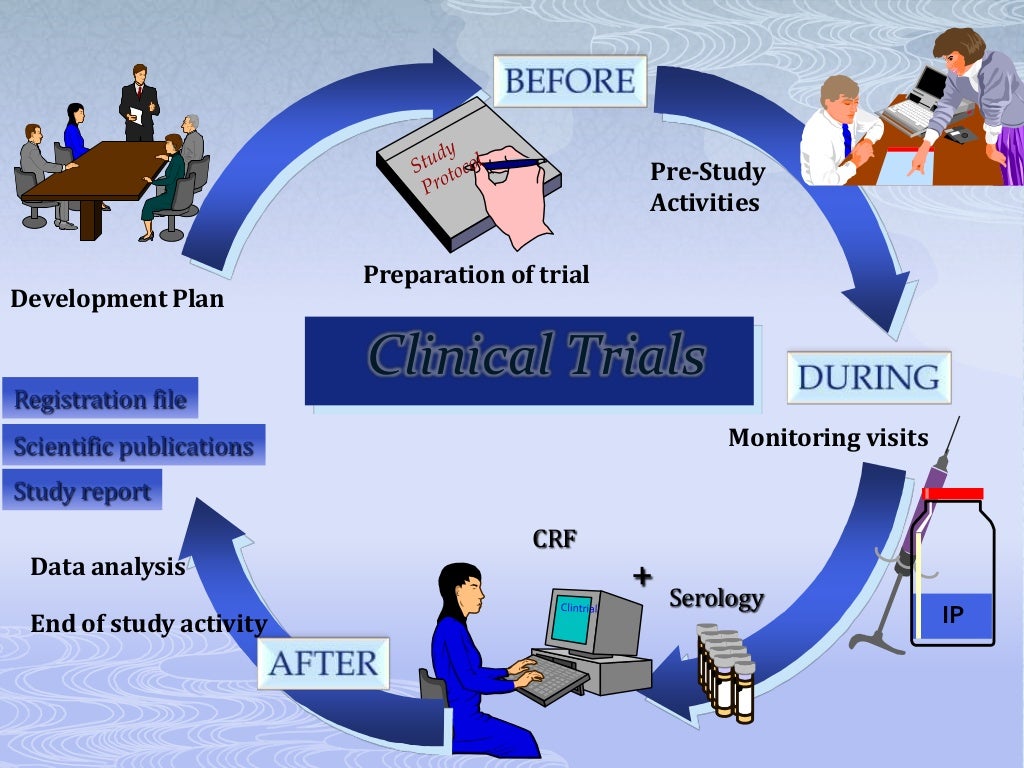
Hiring a contract research organization (CRO) with experience in site negotiations on behalf of sponsors can help expedite contract and budget negotiations. Inexperienced site staff, inadequate budget templates, and extended legal reviews can all delay study start-up. Contracts and BudgetsĬontract and budget negotiation between sites and sponsors can take a considerable amount of time. When site selection is rushed, it often yields inaccurate site information and overly optimistic enrollment projections. Start-up timelines are usually tight, so sponsors and sites need to begin this process as early as possible in order to provide accurate comprehensive assessments. Likewise, the site staff considers the study requirements and performs a feasibility assessment of its own, to estimate the site’s potential enrollment performance and costs. Prior to selecting a site, the sponsor or CRO performs a detailed assessment of the site’s research capabilities, including past clinical trial experience, staff workload, patient population, site facilities and equipment, and qualifications, to determine if the site would be a good fit to participate in the study. Selecting a clinical site is one of the biggest challenges sponsors and CROs face during the start-up process. A well-crafted submission can lead to fewer reviewer questions and faster review times, thus ultimately shortening the start-up timeline. Although counterintuitive, it is beneficial to spend additional time at the outset to ensure the content and quality of the documents to be submitted. Additionally, some countries have rigorous requirements for the content of submission documents and lengthy decision timelines. 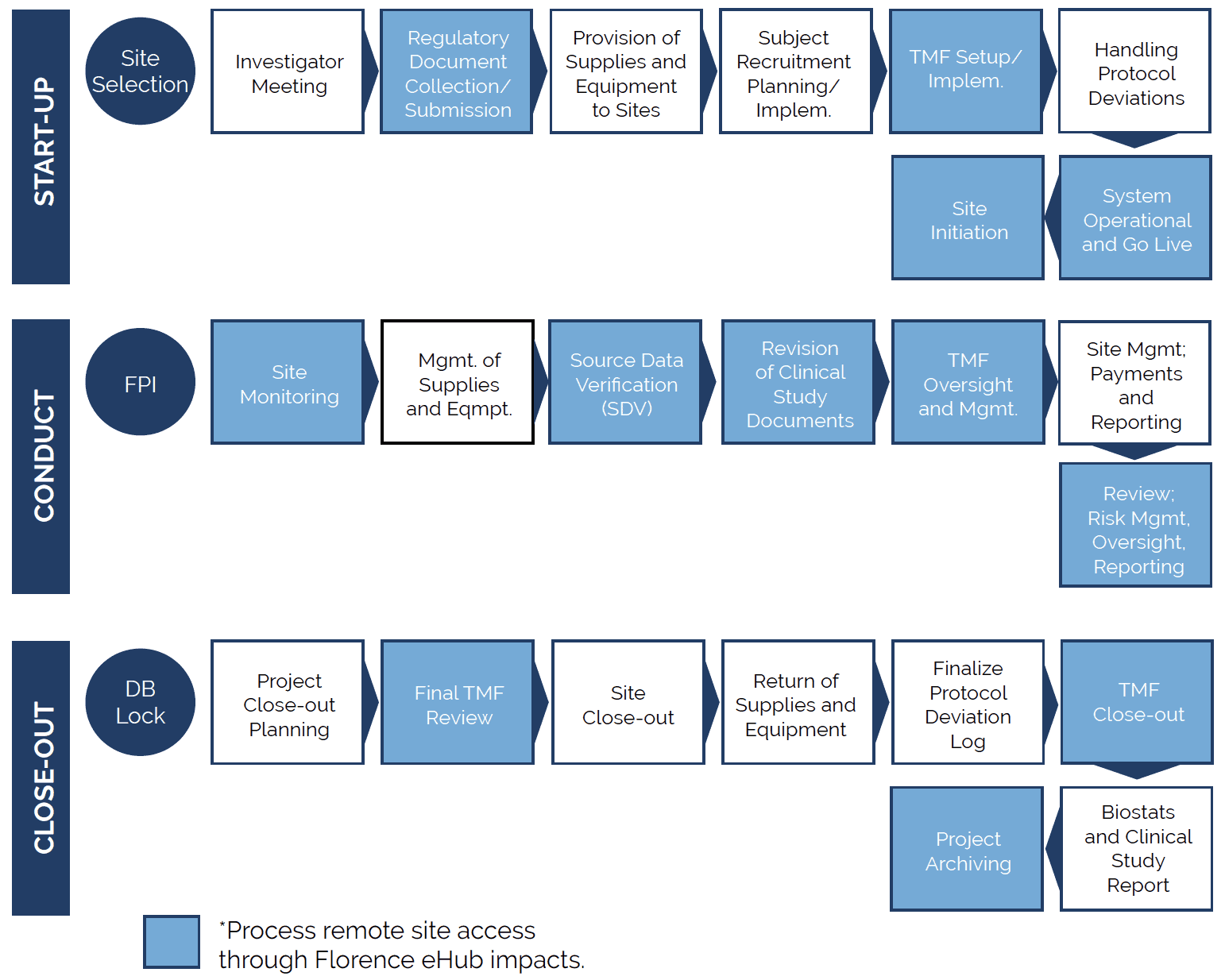
Documents needed for a regulatory submission can be complex and require coordination from team members with different areas of expertise.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
